Bio
Samsung Bioepis’ Enbrel biosimilar reports 270% growth in EU sales
[THE INVESTOR] The European sales of Samsung Bioepis’ first biosimilar Benepali, its copycat version of Amgen’s anti-inflammatory Enbrel, increased nearly threefold in 2017, according to the earnings report from its marketing partner Biogen on Jan. 25.
The biosimilar reported a 269 percent jump in sales to US$370.8 million last year from US$100.6 million in 2016, which is the first year it was launched in Europe.
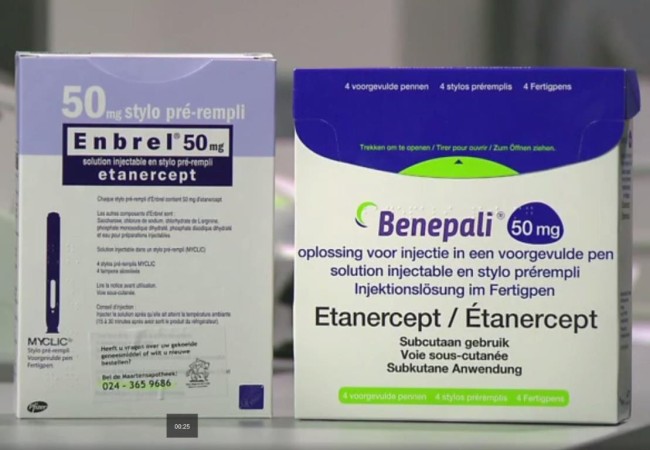 |
Enbrel was the world’s third-best selling drug with global sales of US$8.87 billion in 2016. In February 2016, Samsung Bioepis and its marketing partner Biogen -- which holds a 15 percent stake in Samsung Bioepis -- launched Benepali as the first biosimilar referencing Enbrel to be approved in the EU. During the October to December period in 2017, revenue from Benepali climbed 18.5 percent on-quarter to US$117.6 million.
Meanwhile, Samsung Bioepis’ second biosmilar Flixabi referencing Johnson & Johnson’s rheumatoid arthritis therapy Remicade showed slow market penetration, according to related financial data. Flixabi, which hit the market in August 2016, reported US$9 million revenue in 2017. Remicade had sales of US$6.3 billion last year.
Flixabi is competing with the original drug and other biosimilar product Remsima developed by its local rival Celltrion. Remsima’s first year market share stood at only 1 percent in 2014 to increase to nearly 50 percent this year.
By Park Han-na (hnpark@heraldcorp.com)








