KIST develops new batteries 40% cheaper, stronger than lithium-ion batteries
By Kim Byung-wookPublished : April 19, 2021 - 16:30
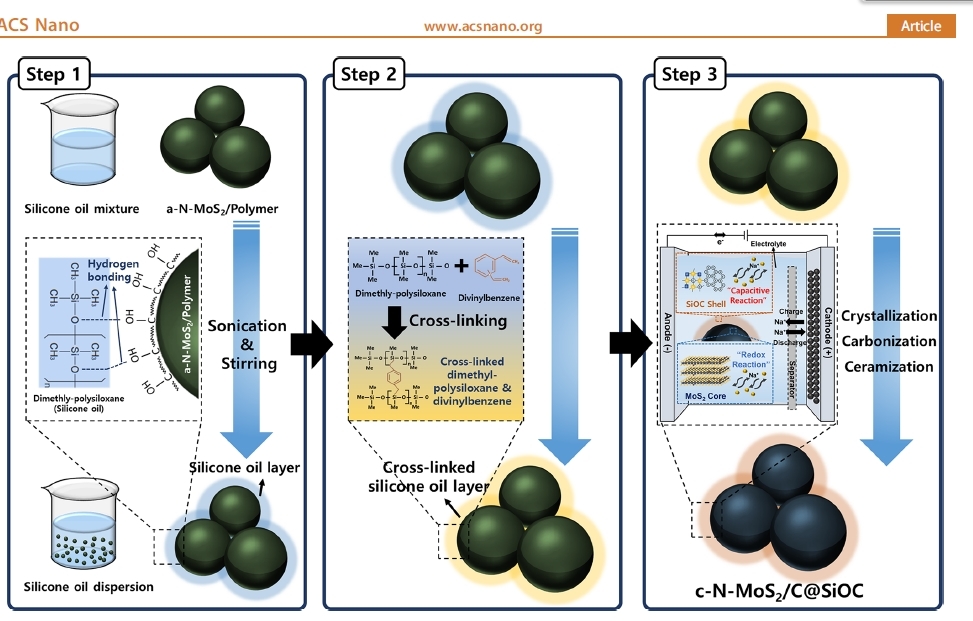
The Korea Institute of Science and Technology on Monday said it has developed sodium-ion batteries that are 40 percent cheaper and can store 1.5 times more charge per gram than conventional lithium-ion batteries.
The batteries use sodium instead of lithium. As sodium is far more common than lithium -- it’s in regular salt -- this brings down the price significantly. However, sodium ions are bigger and heavier than lithium ions, so existing anode materials -- made of graphite and a small amount of silicon -- cannot properly accommodate them.
A team of researchers led by Kim Sang-ok have developed new anode materials customized to handle sodium ions.
Anodes are one of the four key components of lithium-ion batteries along with cathodes, separators and electrolytes. Anodes account for batteries’ stability and lifespan. Lithium-ion batteries are charged and discharged when lithium ions move between cathodes and anodes.
Sodium-ion batteries have the same structure as lithium-ion batteries, but the anodes are made of different materials.
For the new anode materials, Kim used a metallic compound called molybdenum disulfide. This compound can store a lot of electricity, but has a high resistance and is structurally unstable. Kim’s team said that they solved these issues by coating molybdenum disulfide particles with silicon oil, encapsulating those particles inside nano ceramic shells.
Pound-for-pound, these batteries also store more electricity.
“The new molybdenum disulfide anodes applied with silicon oil coating technology exhibit a capacity of 600 milliampere-hours per gram and can maintain the same performance after being charged and discharged rapidly in five minutes 200 times,” a KIST official said. The maximum theoretical capacity of lithium-ion batteries’ graphite anodes is 372 milliampere-hours per gram.
The research has been published in the latest issue of ACS Nano, a global nanoscience journal issued every month.
By Kim Byung-wook (kbw@heraldcorp.com)









![[Hello India] Hyundai Motor vows to boost 'clean mobility' in India](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/25/20240425050672_0.jpg&u=)








