Korea keeps serious vaccine safety issues quiet
Rare complication following AstraZeneca jab leaves woman unable to walk
By Kim ArinPublished : April 19, 2021 - 15:36
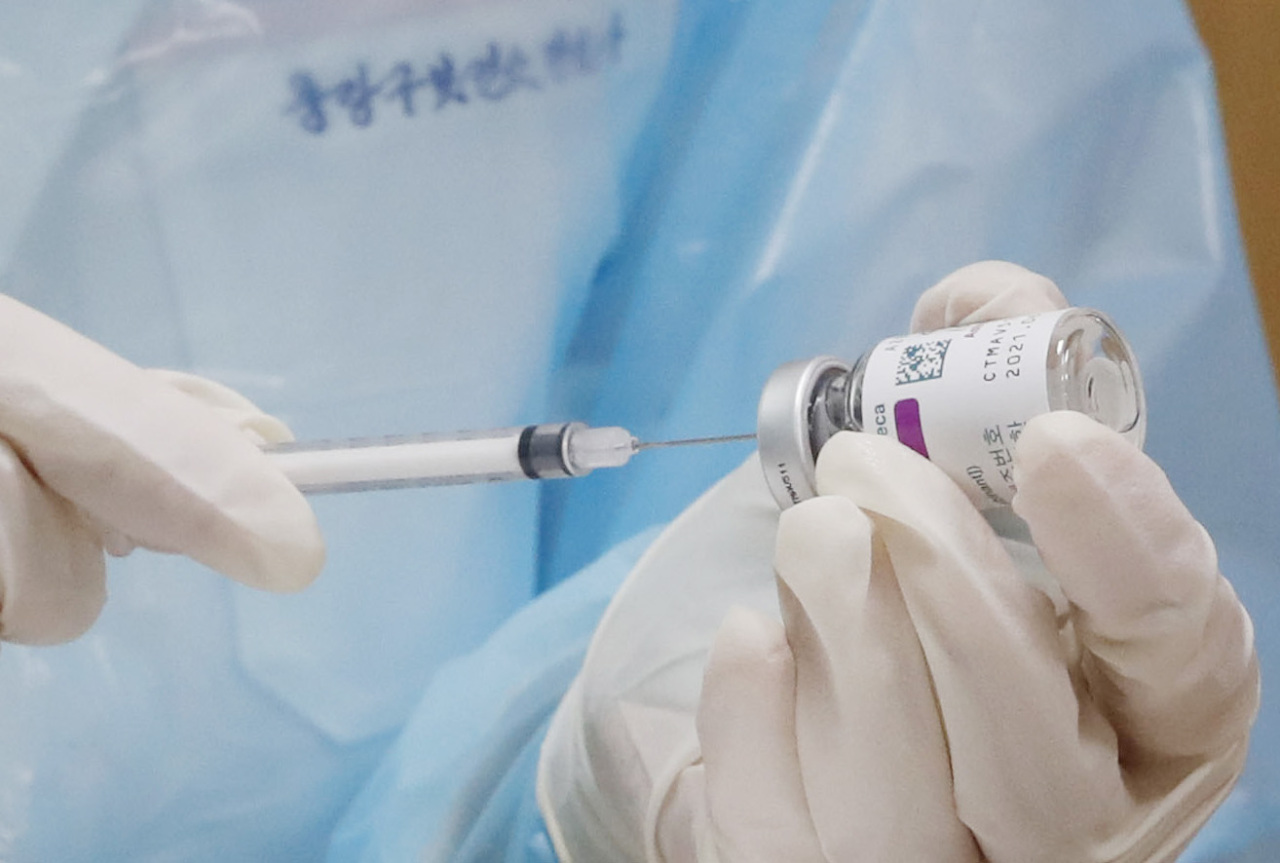
South Korean government officials are failing to share sensitive COVID-19 vaccine safety issues transparently, a move that experts fear may fan public skepticism.
A previously healthy front-line worker in her 40s is hospitalized with a serious neurological condition after receiving AstraZeneca’s COVID-19 vaccine last month, the parliamentary health and welfare committee said Sunday.
The 45-year-old nursing assistant received her first AstraZeneca shot March 12. Soon after, she developed a headache that did not go away and worsened over time. About a week and a half later, her vision began to blur and her temperature rose to 38 degrees Celsius.
She was taken to the hospital March 31, and all four of her limbs have been paralyzed since. She has been diagnosed with an autoimmune disorder called acute disseminated encephalomyelitis, or ADEM, which attacks the central nervous system and causes inflammation in the brain and spinal cord.
Health officials are now being accused of withholding the news from the public despite being alerted to the case as early as March 19.
They were criticized over similar communication delays with previous cases of unusual blood-clotting events in recipients of the AstraZeneca vaccine, waiting two weeks or longer at a time.
In addition, the health authorities did not reveal having used potentially defective syringes to deliver tens of thousands of AstraZeneca vaccinations until press reports brought it to public attention last week. According to the parliamentary committee, they were already aware of the problems with the syringes in the first few days of the vaccination campaign in late February, but did not take any action until March 18.
Epidemiologist Dr. Chun Byung-chul of the Korean Vaccine Society said the Korean public was “entitled to be informed of any emerging safety concerns” as the country strives to vaccinate the majority, if not all, of its population against COVID-19. He called for “greater transparency” from health officials to boost public confidence in the process.
Dr. Paik Soon-young, a virologist at Catholic University of Korea, said the repeated attempts to obscure, rather than come clean with, failures and safety issues surrounding the vaccines constituted “serious neglect” on the part of health officials.
“Such communication failures from the top will cause public trust to falter,” he said.
The Korea Disease Control and Prevention Agency only addressed the severe impairment affecting the nursing assistant in response to a press inquiry during Monday’s televised briefing.
“There will be another round of examinations next month to see what her final diagnosis will be,” said Park Young-joon, who is on the national health agency’s monitoring team in charge of following up on adverse events after immunizations. Whether the vaccine caused her to fall ill was still being investigated.
“One of the primary considerations in causality assessments is if the reported condition is officially listed as the vaccine’s side effect by regulators here or elsewhere. ADEM isn’t one of them,” he said.
Infectious disease professor Dr. Kim Woo-joo of Korea University said ADEM was among the severe nervous system disorders that could be attributed to vaccination, and that its occurrence deserved close attention.
“The background incidence rate is low, at an average of 0.3 cases found per million people a month, and the timeline of the progress of her symptoms after vaccination suggests a possible causal relationship,” he said.
Opposition lawmaker Rep. Suh Jung-sook of the health and welfare committee said the nursing assistant, on top of battling the illness, was being burdened with a monthly hospital bill of several million won. If a cause and effect relationship with the vaccination cannot be determined, all of the costs will continue to be her responsibility.
“As the vaccine rollout expands, there needs to be a way of compensating individuals suffering from suspected vaccine-related injuries,” Suh said in a statement.
So far in Korea, about 1 million people have received at least a single shot of the AstraZeneca vaccine. Five reported a rare form of blood clots, including one person who experienced a clotting disorder in the brain called cerebral venous sinus thrombosis.
By Kim Arin (arin@heraldcorp.com)






![[From the Scene] Monks, Buddhists hail return of remains of Buddhas](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=644&simg=/content/image/2024/04/19/20240419050617_0.jpg&u=20240419175937)








![[From the Scene] Monks, Buddhists hail return of remains of Buddhas](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=652&simg=/content/image/2024/04/19/20240419050617_0.jpg&u=20240419175937)

![[KH Explains] Hyundai's full hybrid edge to pay off amid slow transition to pure EVs](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=652&simg=/content/image/2024/04/18/20240418050645_0.jpg&u=20240419100350)

![[Today’s K-pop] Illit drops debut single remix](http://res.heraldm.com/phpwas/restmb_idxmake.php?idx=642&simg=/content/image/2024/04/19/20240419050612_0.jpg&u=)